This paper includes:
Indeed the International Federation of Clinical Chemistry working group for Standardization of Thyroid Function Tests acknowledged the need for standardization of the fT4 assay.
Have to say, in my facetious moments, I have wonder what the IFCCSTFT group do when they are not stating the obvious.
However, acknowledging an issue and discussing if the same sort of issue could occur due to contraceptive pills or HRT - and in thyroid disease.
The paper also identifies a limit being that if you don’t have an out-of-range TSH, you are unlikely to have an FT4 test!
And it points out that if the assays are biased, they need to use assay-specific reference intervals. Which is like using a ruler that is known to be too short or too long and adding/subtracting every time you measure something. Far better use an accurate ruler?
Pregnancy disrupts the accuracy of automated fT4 immunoassays
in European Thyroid Journal
Authors:
Heleen I Jansen, Antonius E van Herwaarden, Henk J Huijgen, Rebecca C Painter, Jacquelien J Hillebrand, Anita Boelen, and Annemieke C Heijboer
Correspondence should be addressed to H I Jansen; Email: h.jansen@amsterdamumc.nl
DOI: doi.org/10.1530/ETJ-22-0145
Volume/Issue: Volume 11: Issue 6
Article Type: Research Article
Article ID: e220145
Online Publication Date: 31 Oct 2022
Copyright: © The authors 2022
Abstract
Objective
Thyroid hormone measurements are often performed in pregnant women, as hypo- and hyperthyroidism during pregnancy can severely affect the fetus. Serum free thyroxine (fT4) measurements are well known for their analytical challenges, due to low serum concentrations and the subtle equilibrium between free and bound T4 (to thyroid-binding globulin (TBG), transthyretin and albumin). Pregnant women have high TBG concentrations due to an increase in human chorionic gonadotropin (hCG) and estrogen and lower albumin concentrations which change the equilibrium and may affect the validity of fT4 measurements in their samples. As accurate serum fT4 measurements in pregnant women are important for the long-term health of the fetus, we aimed to evaluate the accuracy of several fT4 immunoassays in the serum of pregnant women.
Methods
FT4 was measured in healthy controls and pregnant women using a candidate-reference method (LC-MS/MS) and five commercially available automated immunoassays (Alinity (Abbott), Atellica (Siemens), Cobas (Roche), Lumipulse (Fujirebio) and UniCel DXI (Beckman Coulter)). Method comparisons (Bland Altman plots and Passing and Bablok analyses) were performed.
Results
Serum samples from both healthy controls (n= 30) and pregnant women (n= 30; mean gestational age, 24.8 weeks) were collected. The fT4 immunoassays deviated +7 to +29% more from the LC-MS/MS in serum samples of pregnant women than healthy controls (falsely high).
Conclusions
Our results indicate that immunoassays overestimate fT4 in pregnant women, which might lead to an overestimation of thyroid status. Physicians and laboratory specialists should be aware of this phenomenon to avoid drawing false conclusions about thyroid function in pregnant women.
Keywords: pregnancy; free thyroxine; immunoassay; LC-MS/MS
Open access here:
etj.bioscientifica.com/view...
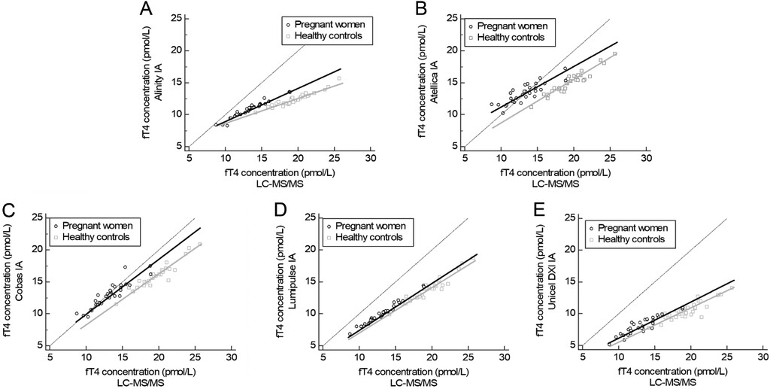
Figure 3
Passing and Bablok regression analyses for the five automated immunoassays in pregnant women and healthy controls. On the x-axis, the fT4 concentrations were measured using LC-MS/MS and, on the y-axis, the fT4 concentrations using the respective immunoassays are shown. (A) Alinity; (B) Atellica; (C) Cobas; (D) Lumipulse; (E) UniCel DXI.