November 2015 my GP informed me he could no longer prescribe Co-proxamol on the 'Named Patient' scheme. Within a month of his decision to end my prescription the recommended alternatives were once again causing me increasing unpleasant gastrointestinal side effects and not providing me with adequate pain relief resulting in me having to lead a life of increased side effects, lack of sleep, and relentless debilitating pain. I previously suffered this situation for 2 out of the 4 years between 2005 and 2008 when all patients were forced off Co-proxamol to try the alternatives prior to me gaining 'Named Patient' status.
My GP was fully aware of the benefits I Received from Co-Proxamol while treating me as a ‘Named Patient’ (2008 – 2015). Throughout this period I Hadn't Suffered Any Gastrointestinal Side Effects and I Maintained a Good Level of Pain Relief.
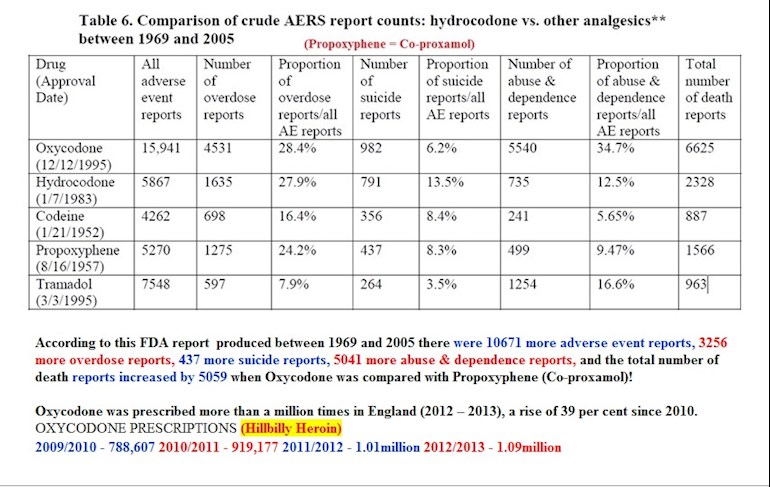
The MHRA deliberately removed the remaining 'Marketing Authorisations' (Licences) for Co-proxamol purely for financial reasons NOT PATIENT SAFETY!
Co-proxamol had been a completely safe (when tolerated and used correctly) drug which had been used by patients for over 50 years, my FOI request [FOI 16/546] outlines the MONETARY IMPACT OF CO-PROXAMOL WITHDRAWAL.
I asked : - (1) What was the monetary loss to MHRA when the Co-proxamol MA’s were cancelled?
MHRA response: - Zero
(2) What was the potential monetary loss to MHRA between 2008 and 2015 for the loss of licence variations and renewals fees?
MHRA response: - This has been calculated to be approximately £100,000.
Alternate Analgesia
How many MA’s were held for OXYCODONE drugs when the phased withdrawal for Co-proxamol was first announced in January 2005? 13
How many MA’s were held in January 2015?151
What was the monetary Gain to MHRA between 2008 and 2015 resulting from any increases in the number of MA's issued? (Including Variation and Renewal Fees).
Approximately £1,550,000. This is the total received in application fees for these new marketing authorisations and does not include any subsequent licence variations or service fees. My attempt at obtaining an answer to this question was cunningly circumvented by them using Section 12 of the Freedom of Information Act. Licence variations and service fees are a continuous income for MHRA which are far more lucrative than the initial marketing authorisations!
Significant new information has appeared through my most recent FOI requests that have been answered. [FOI 16/546] 20th October 2016 and [FOI 17/046] 9th February 2017.
In order to resolve this issue I compromised and requested them to research the first 60 MA's they issued for Oxycodone since 2005 via a new FOI request.
MHRA persuaded the UK government to accept a flawed CSM /MHRA report that had received a meagre response of just 14.7%.
The UK government showed an absolute disregard in respect of ‘DUE DILIGENCE’, and the statistics supplied to CSM /MHRA by Prof Keith Hawton appeared to be dubious at best.
Ministers Directly Responsible For the Lack of Due Diligence
Patricia Hewitt 5 May 2005 – 27 June 2007
Alan Johnson Secretary of State for Health Jun 2007 - Jun 2009
Caroline Flint Minister for Public Health from 2005 to 2007
Dawn Primarolo Minister for Public Health 2007–2009
MHRA HAVE GAINED MANY MILLIONS OF POUNDS AS A DIRECT RESULT OF BANNING CO-PROXAMOL!