As the years go by, we get used to reading about yet more foods, supplements and medicines that affect the thyroid. We see issues like iron and levothyroxine binding to each other in the gut. And the impact of goitrogenic vegetables.
This paper is fascinating because it appears to relate taste and thyroid in a way that few of us would have thought. (Yes – many of us have noticed that being hypothyroid affects perception of taste and our choices of food.)
If I have got the right end of the stick, the idea is something like this:
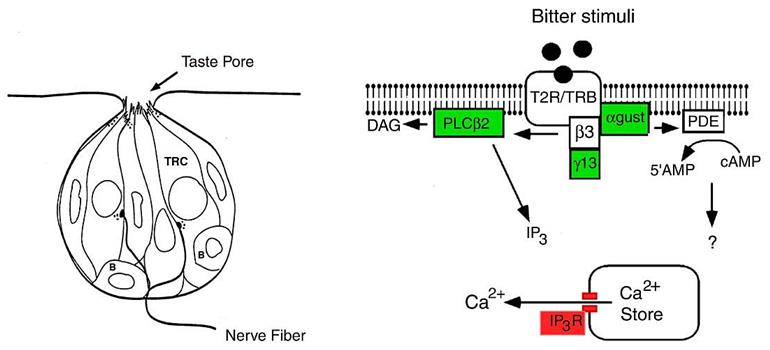
When we eat, our taste receptors respond to the food. Some foods are bitter and some specific taste receptors respond to that. (There are lots of different bitter receptors, this only applies to a few of them.)
Within our thyroids we find some of these same taste receptors. So some substances which are eaten and are perceived as being bitter, then reach the thyroid, where they can trigger the taste receptors. Though they don’t then give us a taste sensation, they do affect T4 and T3 production.
Further, we know that there are people who cannot detect certain bitter flavours. These same people would also not have their thyroids affected by those substances.
FASEB J. 2014 Oct 23. pii: fj.14-262246. [Epub ahead of print]
TAS2R bitter taste receptors regulate thyroid function.
Clark AA1, Dotson CD2, Elson AE3, Voigt A4, Boehm U5, Meyerhof W4, Steinle NI6, Munger SD7.
Author information
Abstract
Dysregulation of thyroid hormones triiodothyronine and thyroxine (T3/T4) can impact metabolism, body composition, and development. Thus, it is critical to identify novel mechanisms that impact T3/T4 production. We found that type 2 taste receptors (TAS2Rs), which are activated by bitter-tasting compounds such as those found in many foods and pharmaceuticals, negatively regulate thyroid-stimulating hormone (TSH)-dependent Ca2+ increases and TSH-dependent iodide efflux in thyrocytes. Immunohistochemical Tas2r-dependent reporter expression and real-time PCR analyses reveal that human and mouse thyrocytes and the Nthy-Ori 3-1 human thyrocyte line express several TAS2Rs. Five different agonists for thyrocyte-expressed TAS2Rs reduced TSH-dependent Ca2+ release in Nthy-Ori 3-1 cells, but not basal Ca2+ levels, in a dose-dependent manner. Ca2+ responses were unaffected by 6-n-propylthiouracil, consistent with the expression of an unresponsive variant of its cognate receptor, TAS2R38, in these cells. TAS2R agonists also inhibited basal and TSH-dependent iodide efflux. Furthermore, a common TAS2R42 polymorphism is associated with increased serum T4 levels in a human cohort. Our findings indicate that TAS2Rs couple the detection of bitter-tasting compounds to changes in thyrocyte function and T3/T4 production. Thus, TAS2Rs may mediate a protective response to overingestion of toxic materials and could serve as new druggable targets for therapeutic treatment of hypo- or hyperthyroidism.-Clark, A. A., Dotson, C. D., Elson, A. E. T., Voigt, A., Boehm, U., Meyerhof, W., Steinle, N. I., Munger, S. D. TAS2R bitter taste receptors regulate thyroid function.
© FASEB.
KEYWORDS:
G protein-coupled receptor; T2R; iodide; thyrocyte; thyroxine
PMID: 25342133 [PubMed - as supplied by publisher]
ncbi.nlm.nih.gov/pubmed/253...
(As so often, the full paper is not available.)
A bit of background on taste receptors (usual warnings that Wiki isn’t perfect):
en.wikipedia.org/wiki/Taste...
scholarblogs.emory.edu/evol...
Rod