Published on line July 11, 2019, Accepted by JCO May 31 2019
Dr P Hillmen
We have some members who are in this important CLARITY Study in UK.
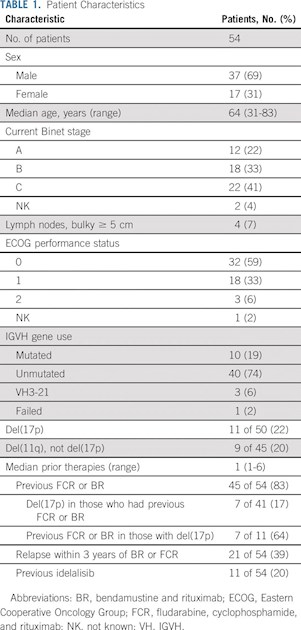
CLARITY is a phase II trial that combined ibrutinib with venetoclax in patients with relapsed or refractory CLL. The primary end point was eradication of MRD after 12 months of combined therapy.
In 53 patients after 12 months of ibrutinib plus venetoclax, MRD negativity (fewer than one CLL cell in 10,000 leukocytes) was achieved in the blood of 28 (53%) and the marrow of 19 (36%). Forty-seven patients (89%) responded, and 27 (51%) achieved a complete remission. After a median follow-up of 21.1 months, one patient progressed, and all patients were alive.
More here: ascopubs.org/doi/full/10.12...
Jackie