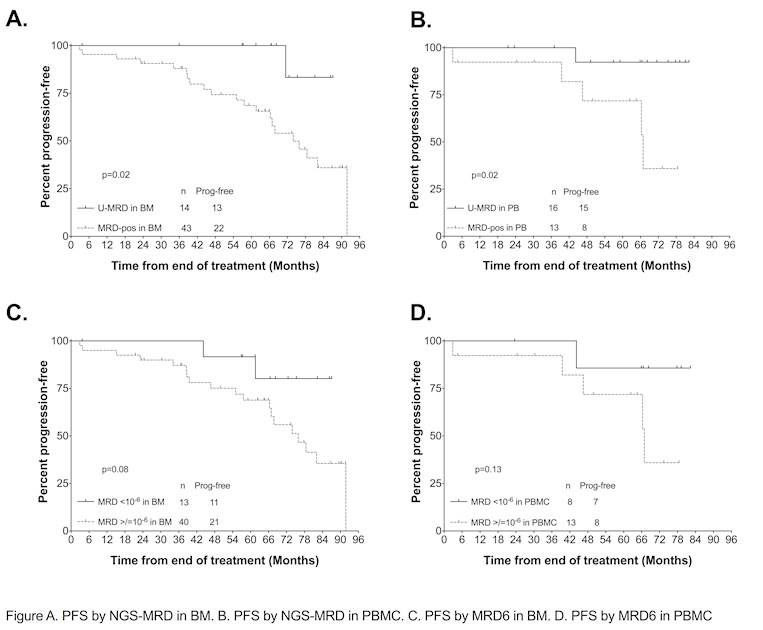
U-MRD6 = Undetectable-Minimal Residual Disease (U-MRD6) (10-6 sensitivity)
Discussion
The majority of pts with BM U-MRD4 after first-line FCR were MRD6+ and these patients had shorter PFS; MRD analysis with a more sensitive assay may therefore more accurately assign prognosis. Not accounting for sensitivity, a higher proportion of BM than PBMC samples were MRD+. Plasma analysis was uninformative as all pts MRD+ in plasma were MRD+ in simultaneous BM or PBMC samples.
Defining prognostically-relevant thresholds for MRD using more sensitive methodology is important, particularly if U-MRD is used a surrogate for PFS in clinical trials or as an endpoint for treatment cessation. MRD6 may become increasingly relevant if venetoclax-based regimens and the addition of novel agents to chemoimmunotherapy achieve U-MRD6 in more patients than FCR. In this retrospective study, a number of samples did not achieve 10-6 sensitivity; optimization of sample collection is important to achieve 10-6 sensitivity in most pts. Finally, additional studies will be required to determine the risk for and kinetics of relapse in patients with low-level MRD6+.