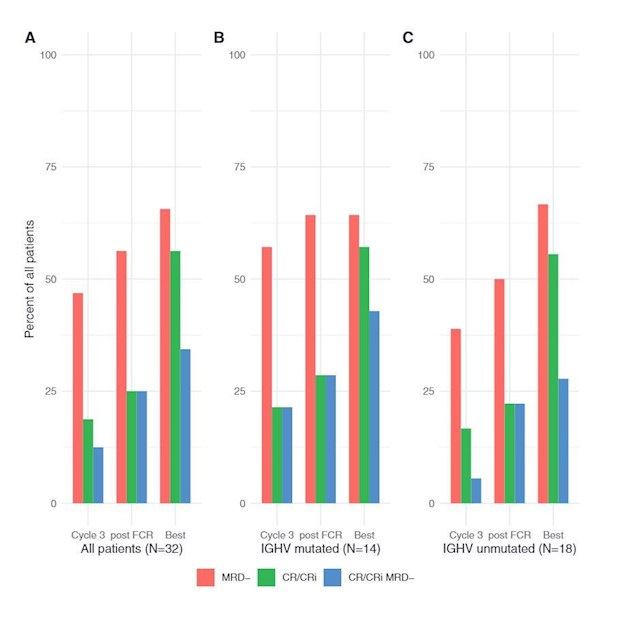
"In summary, our phase 1b/2 study identified a recommended phase 2 dose of duvelisib 25 mg bid when given in combination with FCR in younger CLL patients receiving frontline therapy. We observed deep responses, including achievement of BM-uMRD in about two-thirds of patients,irrespective of IGHV mutation status; however, while the regimen is certainly active, the 3-year PFS of 73% does not appear significantly superior to historical results with FCR alone. Immune-mediated toxicities and infectious complications were relatively common, but with active intervention were manageable for most patients. If DFCR were to be studied further, strategies that include a shorter course of FCR and other approaches to mitigate toxicity while maintaining efficacy would need to be explored."
nature.com/articles/s41375-...
For comparative reference, in the (E1912) phase 3 trial for untreated younger patients, the 3-year PFS was 89% for IR and 73% for FCR.